Emiliano Panieri, Debora Romoli
Over the last hundred years, global production of chemicals has increased exponentially, going from 1 million tons in 1930 to several hundred million tons today. The European Union (EU) is the world's second largest producer after China and it is estimated that there are more than 100,000 chemicals on the European market. Italy, with a production value of over 65 billion euros, is the third European producer, after Germany and France, and the tenth globally. There are approximately 2,800 chemical companies active in Italy and they employ over 113,000 people, but the use of chemical products affects all production sectors. European regulation of chemicals is probably the most ambitious in the world, its aim is to ensure a high level of protection of human health and the environment. The main reference standards are the REACH Regulation ( Registration, Evaluation, Authorization and Restriction of Chemicals ) and the CLP Regulation ( Classification, Labeling and Packaging ). Safety in the management of chemical substances is primarily the responsibility of the companies that produce, import or use them. ECHA ( European Chemicals Agency ) and the competent authorities of the EU Member States carry out a control activity on the obligations of companies and can intervene with specific measures if it is demonstrated that the risk of the substances for humans and the environment is not adequately controlled. The indicator, through the monitoring of regulatory processes, provides the elements to evaluate progress in achieving the safety objectives established by the legislation.
Chemicals in Europe are regulated through comprehensive strategies and sector-specific regulations. The REACH regulation ( Registration, Evaluation, Authorization and restriction of CHemicals ) applies to all sectors dealing with such substances throughout their life cycle. Its aim is to ensure a high level of protection of human health and the environment, promoting the competitiveness and innovation of the European chemical industry, with a particular commitment to the use of alternative methods to animal testing. The CLP Regulation ( Classification, Labeling and Packaging ) ensures that the hazards of chemicals are clearly communicated through classification and labeling throughout the supply chain. The indicator measures the effectiveness of the main European regulatory processes responsible for the safety of chemical substances: registration, evaluation of registration dossiers, evaluation of priority substances, risk management measures adopted at European level (harmonized classification, restrictions, authorisation).
Monitor the ability of the legislation to achieve the fundamental objective of raising the level of safety in the management of chemical substances in all their life phases.
More than twenty years after the first strategic approach for the management of chemicals in Europe (White Paper: Strategy for a future chemicals policy), the European Union has outlined a new long-term strategy on chemicals. In line with the Green Deal , the European strategy aims to create a toxic-free environment, in which chemicals are produced and used in a way that maximizes their contribution to society, while simultaneously avoiding harm to the environment and to current and future generations. In this regard, the European Union has one of the most complete and protective regulatory frameworks regarding chemical substances, which has around forty instruments, including the REACH and CLP regulations, the two large regulations that act horizontally and regulate all chemical substances, and a series of sectoral regulations that regulate specific types of substances and products (phytosanitary products, biocides, cosmetics, etc.).
Regulation (EC) no. 1907/2006 concerning the registration, evaluation, authorization and restriction of chemical substances (REACH), which came into force in 2007, aims to fill knowledge gaps on substances, to promote the replacement of those most worrying for human health and the environment with safer alternatives and to make risk management more efficient. This is done primarily by placing the burden of safety on companies that have the obligation to manufacture, place on the market or use substances that do not cause harm to human health or the environment.
Regulation (EC) no. 1272/2008 on the classification, labeling and packaging of dangerous substances and mixtures (CLP), which entered into force in January 2009, is based on the United Nations Globally Harmonized System of Classification and Labeling of Chemical Substances (GHS) and aims to ensure a high level of health and environmental protection, as well as the free movement of substances, mixtures and articles.
In particular, to guarantee chemical safety, the REACH regulation establishes a single system for risk management, which provides:
- the registration of all substances produced and imported above 1 tonne per year;
- the evaluation of registration dossiers;
- the evaluation of substances considered priorities in terms of quantity (tonnage) and dangerous characteristics;
- the application of risk management measures at community level, such as restriction and authorisation, for those substances which present unacceptable levels of risk to human health and/or the environment.
Based on the experience gained in the first years of implementation of the REACH and CLP regulations, ECHA has developed since 2016 an integrated regulatory strategy that brings together the various regulatory processes. The strategy provides a clear and coherent basis for achieving the objectives of the Regulations and contributing to the achievement of the United Nations Sustainable Development Goals on chemicals. The strategy aims to:
- efficiently select substances or groups of substances of potential concern and generate the information necessary to assess their safety, so that any remaining concerns can be addressed through the most appropriate regulatory risk management tool;
- ensure appropriate and timely intervention by all actors (ECHA, Member States, European Commission and Industry), under the different REACH and CLP processes, so that chemicals of concern are addressed adequately and as early as possible;
- provide stakeholders and the public with certainty that registering companies comply with the information requirements of the REACH Regulation, promoting better communication on safe use in the supply chain.
EC, Regulation (EC) n. 1272/2008 relating to the classification, labeling and packaging of dangerous substances and mixtures (CLP)
CEFIC, 2025, Facts and Figures of the European Chemical Industry
ECHA, https://echa. europa. eu/it/home
ECHA, 2025a, Annual Report 2024, ISBN 978-92-9468-469-1
ECHA, 2025b, Evaluation progress-ECHA-substance evaluation 2012-2024 (https://echa. europa. eu/it/progress-in-substance-evaluation)
Federchimica, 2025, The chemical industry in figures 2025
EU, Towards a toxic-free environment, COM(2020) 667 final
Absence of reference values
None
CEFIC - European Chemical Industry Council
ECHA - European Chemicals Agency
Federchimica
CEFIC 2025_Facts_and_Figures_Data files xls ( https://cefic. org/resources/data-files-xls-2024-cefic-facts-and-figures/)
Federchimica the chemical industry in numbers - 2025 ( https://www. federchimica. it/industria-chimica-in-cifre )
REACH registration statistics, Date as of: 31/12/2024 ( https://echa. europa. eu/it/registration-statistics )
ECHA, Substance Evaluation table ( https://echa. europa. eu/it/information-on-chemicals/evaluation/community-rolling-action-plan/corap-table )
ECHA, Classification and Labeling Inventory Database ( https://echa. europa. eu/it/information-on-chemicals/cl-inventory-database )
ECHA, List of substances of very high concern candidates for authorization ( https://echa. europa. eu/it/candidate-list-table )
ECHA, List of restrictions ( https://echa. europa. eu/it/substances-restricted-under-reach )
ECHA, List of Authorizations ( https://echa. europa. eu/it/authorisation-list )
ECHA, Evaluation progress ( https://echa. europa. eu/it/overall-progress-in-evaluation )
ECHA, Registry of restriction intentions until outcome ( https://echa. europa. eu/it/registry-of-restriction-intentions )
ECHA, Annual Report 2024, ISBN: 978-92-9468-469-1 ( https://echa. europa. eu/documents/10162/7362407/annual_Report_2023_en. pdf/ca16f5a4-a8ae-ac22-21f1-57af476f0bb1? t=1713865234883)
EU, National
2008-2024
The processing procedure involves the collection of data from the reference source and their representation as punctual or cumulative data, respectively when associated with a single year or instead with a specific time interval. In some cases, the representation/processing required the application of calculation procedures and/or Boolean operators.
The regulatory processes responsible for the safety of substances are:
- Registration of substances produced or imported and placed on the European market above 1 tonne per year
- Evaluation of substance registration dossiers
- Evaluation of priority substances
- Risk management measures adopted at European level (harmonised classification, restrictions, authorisation)
By registering, companies submit to the European Chemicals Agency, ECHA ( European Chemicals Agency ), a registration dossier with information on the properties, uses of the substances and the methods implemented to control the risks. The classifications carried out by companies (self-classifications) are communicated to ECHA, which makes them accessible, together with the harmonized ones, through the Classification and Labeling Inventory. The inventory is the main reference for harmonized classification and a tool for the convergence of self-classifications, encouraging companies to agree on a shared classification. The information is also used by Member States to identify potentially dangerous substances. ECHA carries out checks on registration dossiers, evaluating the completeness and adequacy of the information, and examining testing proposals in order to avoid unnecessary tests on animals.
The joint action of the REACH and CLP regulations guarantees that every year a certain number of substances are managed at a regulatory level in order to guarantee greater safety for humans and the environment. However, due to the very high number of chemical substances present on the European market, the evaluation and subsequent regulatory processes carried out on an annual basis can only cover a part of these substances.
From the beginning of the substance evaluation process (2012) until 2024, evaluations of 327 substances were started (ECHA, 2025b), to identify the most problematic potential hazard characteristics associated with relevant exposure conditions. So far, a high percentage of assessments (70%) have led to requests for additional information. From 2009 to 2024, ECHA published over 4,400 decisions covering approximately 15,000 requests for information in the context of compliance checking of registration dossiers, examination of testing proposals and substance evaluation (Table 3). Since the entry into force of the REACH regulation, 242 substances have been included in the list of candidate substances for the authorization process (Figure 6) and 53 restriction proposals have been submitted (Table 1). Finally, 625 opinions on the harmonized classification and labeling proposals were adopted by ECHA's Risk Assessment Committee (Table 2).
Over the years the number of regulated chemical substances is growing significantly and therefore it can be assumed that the trend relating to chemical safety is positive. However, it is very difficult to say when the ambitious goal of regulating all chemicals of concern that are marketed in Europe will be achieved.
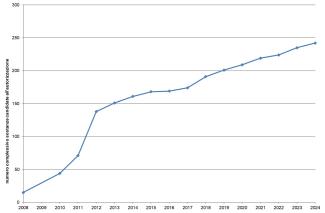
The safety of chemicals in terms of their impact on human and environmental health is increasing overall, as a result of the growing commitment at European level in the application of risk management measures in compliance with the REACH and CLP regulations. In particular, this led to a progressive increase both in the number of substances registered from 2009 to 2024 (Figure 2) and in the regulatory processes responsible for safety.
| Data |
|---|
Headline
Produzione chimica mondiale (anno 2024) Data source
Federchimica industria chimica in cifre 2025 Cefic Facts and Figures 2025 |
Headline
numero sostanze registrate (2024) Data source
Elaborazione ISPRA su dati ECHA (aggiornati al 31-12-2024) |
Headline
Figura 3: numero registrazioni per paese (2024) Data source
Elaborazione ISPRA su dati ECHA (aggiornati al 31-12-2024) |
Headline
numero sostanze registrate per paese (2024) Data source
Elaborazione ISPRA su dati ECHA (aggiornati al 31-12-2024) |
Headline
Motivi di preoccupazione per le sostanze inserite nelle valutazioni CoRAP fino al triennio 2025-2027 Data source
Elaborazione ISPRA su dati ECHA (aggiornati al Novembre 2025) |
Headline
sostanze estremamente preoccupanti candidate all'autorizzazione (2008-2024) Data source
Elaborazione ISPRA su dati ECHA (aggiornati al dicembre 2024) |
Headline
Table 1: Restriction proposals and decisions (2010-2024) Data source
ISPRA processing of ECHA data |
Headline
Table 2: Number of CLH opinions by year (2009-2024) Data source
ISPRA processing of ECHA data |
Headline
Table 3: Trend of evaluation processes from 2009 to 2024 Data source
ECHA |
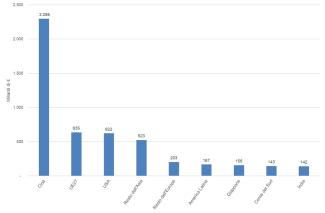
Thousands of chemicals are produced, consumed, and traded worldwide. The chemical sector plays a significant role in the global economy with growing development trends, although still marked by the 2008 global economic crisis and the 2020 health crisis. Global chemical industry production was valued at €5,031 billion in 2024. With a 46% share, China is the world's leading producer, accounting for €2,298 billion. The European chemical industry, with €635 billion in turnover and a 13% share, is the world's second-largest producer (Figure 1).
Italy, with a production value of over €65 billion in 2024, is confirmed as the third-largest European producer after Germany and France, and the tenth globally. There are more than 2,800 chemical companies active in Italy, employing over 113,000 people (Federchimica, 2025). Italian chemical production primarily develops across three sectors:
Although activities are distributed across the national territory, the highest concentration is in Northern Italy, with nearly 78% of the sector's employment (approximately 40% in Lombardy), compared to about 12% in Central Italy and 10% in Southern Italy (Federchimica, 2025).
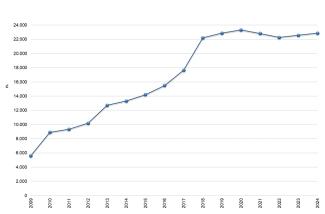
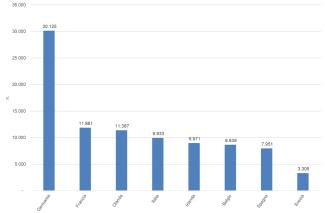
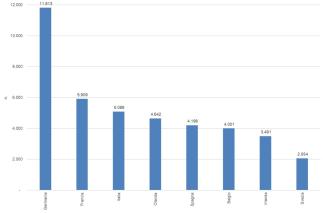
The registration of chemical substances produced and imported above one tonne/year is the first and main obligation of the REACH Regulation to ensure the safe use of chemicals. From the entry into force of REACH until December 31, 2024, 108,391 registration dossiers for 22,872 substances (Figure 2) have been submitted to the European Chemicals Agency. The majority of registrations were made by companies located in Germany (28%), France (11%), and the Netherlands (11%). Italy, with 9,933 registrations (9%) relating to 5,086 substances, ranks fourth among Member States (Figures 3 and 4).
ECHA, using the information provided in the registration dossiers, maintains a database that is also useful in other regulatory processes, with the aim of both defining appropriate risk management measures and making information on chemical substances available to the public. This information serves as the starting point for companies to prepare Safety Data Sheets and to communicate conditions for the safe use of substances throughout the supply chain. It is therefore essential to ensure the quality of registration dossiers, ensuring that the information complies with regulatory requirements and is easily accessible. For this reason, the Agency exercises control over the registration dossiers submitted by companies, carrying out two types of assessment:
Between 2009 and 2024, ECHA, with the support of Member States, conducted 4,335 compliance checks and examined 2,480 testing proposals. In cases of non-compliance for CCHs, and in all cases for TPEs, a request for further information is sent to the Registrant. The compliance check aims to verify:
Compliance checks are primarily targeted at substances (or groups of substances) that raise potential concerns based on hazard or exposure, where improving the quality of information can lead to a significant increase in safety.
Testing proposals must be submitted for registrations exceeding 100 tonnes/year for the information required in Annexes IX and X of REACH. The Agency evaluates all proposals with the goal of avoiding unnecessary testing, particularly on vertebrate animals, which may only be carried out as a last resort when absolutely necessary, avoiding duplication. Since 2009, ECHA has adopted 2,590 decisions on compliance checks concerning 10,987 information requirements where data gaps were identified. Furthermore, the Agency has adopted 1,681 decisions on testing proposals concerning 3,293 information requirements for which testing was originally proposed (Table 3).
Registered substances that are prioritized due to volume and hazard characteristics undergo a more in-depth evaluation by the Competent Authorities of the Member States under the Community Rolling Action Plan (CoRAP), coordinated by ECHA over a three-year period. Priority criteria for selecting substances take into account hazards, exposure, and total volumes placed on the market. The primary purpose of the evaluation is to confirm or dismiss the identified 'initial grounds for concern' and, if necessary, identify the most appropriate risk management measures. The evaluation involves an in-depth review of the chemical safety reports produced by the registering companies, requesting additional information if needed, and includes the drafting of an evaluation report. In the twenty years of application of previous legislation, approximately 140 substances were evaluated.
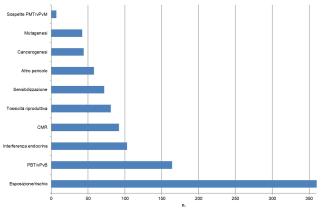
Under the REACH Regulation, between 2012 and 2024, evaluations were initiated for 327 substances to identify potential hazard characteristics such as carcinogenicity, mutagenicity, reproductive toxicity, sensitization, PBT (Persistence, Bioaccumulation and Toxicity), PMT (Persistence, Mobility and Toxicity), and endocrine disruption, associated with relevant exposure conditions (ECHA, 2025b). To date, a high percentage of evaluations (70%) has led to requests for additional information, confirming the validity of the initial grounds for concern for substances included in the CoRAP process through the 2025-2027 period (Figure 5).
After assessing the available or requested information, the Member States responsible for the evaluation concluded whether 254 substances constituted a risk to human health and/or the environment and decided how to use the information on those substances for regulatory risk management. Member States concluded that for 109 substances, no further EU-level regulatory follow-up was justified, while for 145 substances, further EU-level regulatory follow-up was necessary (harmonized classification and labeling, identification as a substance of very high concern, restriction, or other EU-level risk management measures such as an EU-wide occupational exposure limit) (ECHA, 2025b).
The evaluation process plays a fundamental role in the implementation of REACH. In particular, it was essential for achieving the objective set by the 'SVHC Roadmap' to include all 'Substances of Very High Concern' (SVHC) present on the market in the Candidate List for authorization by the end of 2020. The work initiated with the roadmap continues today with the goal established by the Integrated Regulatory Strategy to prioritize risk management and/or data generation for all registered substances by 2027. Substances on the Candidate List may be moved to the list of substances subject to authorization (Annex XIV of REACH). Once included in this list, companies will not be allowed, as of a certain date, to place these substances on the market or use them unless they have been authorized to do so. Through the authorization procedure, the REACH Regulation ensures that SVHCs are progressively replaced by less dangerous substances or technologies. Since 2008, 242 substances or groups of substances have been identified as SVHCs and included in the Candidate List (Figure 6), while 59 substances have been included in Annex XIV. Annex XVII of REACH, instead, contains all the restrictions adopted, including those from the previous Directive 76/769/EEC. Restrictions are, therefore, a risk management measure that has been active for forty years. As of December 31, 2024, Annex XVII includes 79 entries relating to restricted substances or families of chemical substances, some of which are very numerous. Since the entry into force of the REACH regulation, the number of restriction proposals submitted by Member States and ECHA has been 53 (Table 1). From 2009 to 2024, ECHA's Risk Assessment Committee adopted 625 opinions on harmonized classification and labeling (CLH) proposals (Table 2), while the Inventory contains classification and labeling information for approximately 350,000 substances.